A new gene-editing treatment called CRISPR is showing that it can correct some inherited diseases for good.

A revolutionary gene-editing tool called CRISPR is now showing real promise in treating—and in some cases functionally curing—genetic diseases once thought untreatable. Recent clinical trials have demonstrated that the technique can precisely correct DNA mutations responsible for disorders like sickle cell disease and beta thalassemia. With the first CRISPR-based therapy now approved for patients, scientists say we are entering a new era of medicine—one where targeted gene repair could permanently eliminate the root cause of inherited illnesses.
1. CRISPR Works Like a Molecular Pair of Scissors
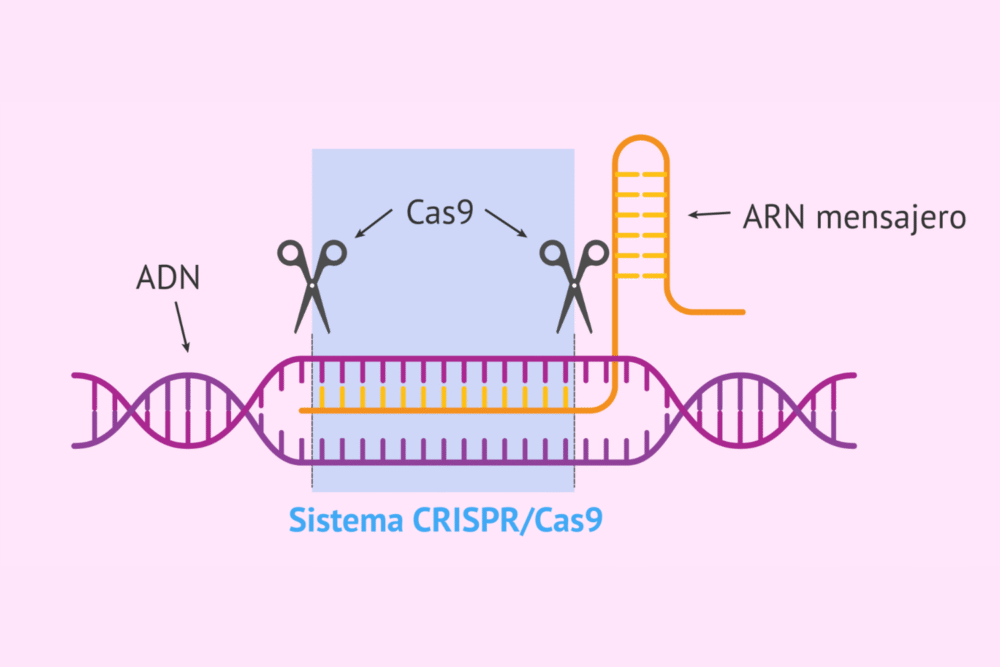
CRISPR, short for Clustered Regularly Interspaced Short Palindromic Repeats, allows scientists to target and modify specific sections of DNA. The system uses a protein called Cas9, guided by RNA, to locate faulty genes and cut them at precise points. Once the cut is made, the cell’s natural repair mechanisms can replace or correct the defective sequence.
This process makes it possible to fix mutations directly at their source, unlike traditional treatments that only manage symptoms. The technology was first adapted for gene editing in 2012 and has since transformed biological research and experimental medicine.
2. First CRISPR Therapy Approved for Human Use

In late 2023, health regulators in the United Kingdom and United States approved the world’s first CRISPR-based treatment, known as Casgevy. The therapy targets sickle cell disease and beta thalassemia—two blood disorders caused by a single faulty gene that affects hemoglobin production.
Casgevy works by editing the patient’s own stem cells to reactivate a dormant form of hemoglobin, allowing healthy red blood cells to form. After the edited cells are infused back into the body, patients have shown sustained symptom relief, marking a major milestone in genetic medicine.
3. The Treatment Shows Remarkable Early Success

Clinical trial data for Casgevy are encouraging. Nearly all participants with severe sickle cell disease or beta thalassemia experienced dramatic improvement after receiving the treatment, with many remaining symptom-free for more than a year.
Patients who previously required frequent blood transfusions no longer need them. While long-term monitoring continues, early results suggest CRISPR therapy can deliver durable, possibly lifelong benefits. Researchers caution, however, that these therapies are still new and must be followed over time to confirm their lasting safety and effectiveness.
4. How CRISPR Differs from Conventional Gene Therapy
Traditional gene therapy introduces new genetic material into cells but doesn’t always correct the faulty gene itself. CRISPR, by contrast, edits the existing DNA directly, providing a more targeted and potentially permanent solution.
This precision reduces the risk of unintended side effects, such as random gene insertions that could disrupt other important DNA functions. Because CRISPR acts like an exact molecular tool, it offers scientists far greater control and accuracy compared to older gene-editing techniques like zinc-finger nucleases or TALENs.
5. Other Diseases Are Now in Clinical Trials

Researchers are testing CRISPR for a growing list of genetic conditions beyond blood disorders. Trials are underway for muscular dystrophy, cystic fibrosis, and inherited blindness, as well as some cancers and rare metabolic syndromes.
Each trial tailors the CRISPR approach to the specific mutation involved. While the early focus has been on single-gene diseases, scientists are now exploring complex conditions influenced by multiple genes. The goal is to expand CRISPR’s reach from rare disorders to more common illnesses that affect millions worldwide.
6. The Technology Is Also Being Used Inside the Body
Most CRISPR treatments so far have been performed “ex vivo,” meaning cells are edited outside the body and then reintroduced. However, recent studies are testing “in vivo” delivery, where CRISPR components are injected directly into patients to make corrections on site.
In 2021, researchers successfully used this method to reduce defective protein production in people with a rare liver disease called transthyretin amyloidosis. The results proved that CRISPR can work safely inside the human body, opening new doors for treating conditions that affect tissues like the heart, lungs, and brain.
7. Ethical Oversight Remains Crucial
Because CRISPR can permanently change human DNA, it raises complex ethical issues—especially concerning germline editing, which would affect future generations. International guidelines currently prohibit using CRISPR to alter embryos intended for pregnancy after the 2018 case in China where edited embryos were born.
Today’s CRISPR therapies target only somatic cells, meaning changes aren’t passed to offspring. Scientists and regulators stress that strict oversight and transparency are essential to ensure the technology is used safely, ethically, and equitably across the globe.
8. Accessibility and Cost Pose Major Challenges

CRISPR-based therapies are still extremely expensive—currently costing several million dollars per patient. The manufacturing process, hospital stays, and conditioning treatments make them accessible only in specialized centers.
Researchers are working to simplify and lower the cost of production through automation and improved gene-delivery systems. As technology scales and becomes more efficient, prices could drop significantly, allowing broader access and transforming genetic medicine from experimental to mainstream care.
9. Potential Risks Are Still Being Studied

Although CRISPR has shown high precision, there’s always a risk of “off-target” edits—unintended DNA changes that could cause harm. Scientists are improving algorithms and screening tools to detect and prevent such errors before therapy reaches patients.
Another concern is the possibility of immune reactions to the Cas9 enzyme, which is derived from bacteria. So far, trials haven’t shown major safety issues, but researchers continue to monitor participants closely to ensure no delayed side effects emerge over time.
10. The Future May Include One-Time Cures
If CRISPR therapies continue to prove safe and effective, they could offer one-time, lasting treatments that replace lifelong medication. For conditions like sickle cell disease, that means patients could be cured with a single infusion instead of years of hospital visits and transfusions.
This shift would represent a fundamental change in medicine—treating genetic disease at its root rather than managing symptoms. Many researchers compare the potential of CRISPR to the invention of antibiotics or vaccines in terms of its long-term global impact.
11. A New Era of Medicine Is Beginning

CRISPR’s success marks the dawn of precision medicine, where treatments are tailored to an individual’s unique genetic code. By understanding and editing DNA directly, scientists can target diseases with unprecedented accuracy and potentially eliminate them altogether.
While more work lies ahead, the first approved CRISPR therapy proves that gene editing has moved from theory to clinical reality. For millions living with inherited disorders, this breakthrough offers not just hope—but a glimpse of a future where genetic diseases could finally be left behind.
